KLK8 (Gene)
Analyze
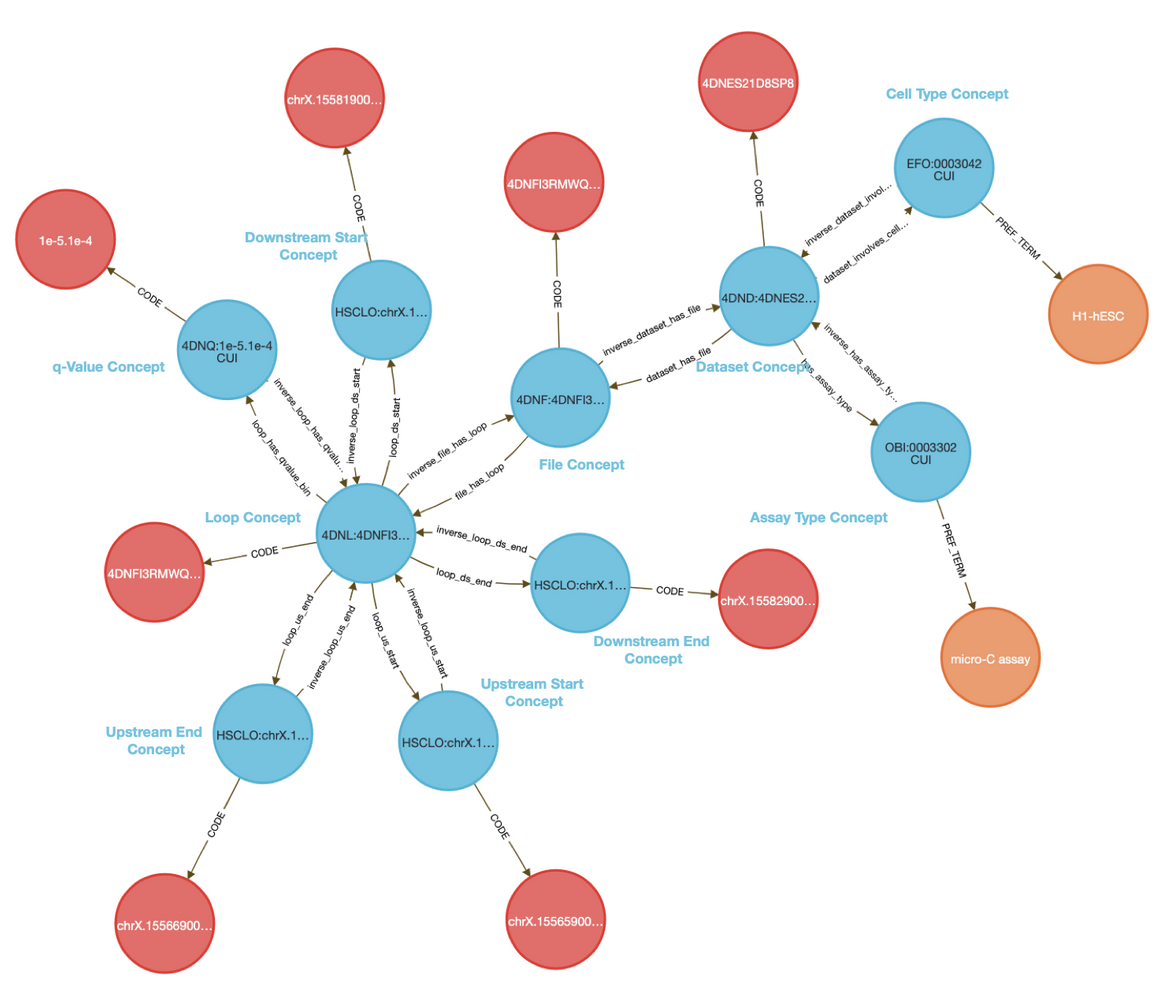
The CFDE Data Distillery Knowledge Graph contains entities and relationships across the CFDE. View KLK8's neighborhood in the knowledge graph.
View Gene-Centric information about the gene from a pre-built PWB workflow. View the workflow with KLK8.

The CFDE Gene Centric Appyter Resolves and Displays Gene-Centric information from CFDE APIs. Execute the Appyter using KLK8.

The Gene and Drug Landing Page Aggregator (GDLPA) finds links to primary and secondary source information from CFDE and other resources. Discover landing pages for KLK8.

The Playbook Workflow Builder helps you interactively construct workflows leveraging CFDE APIs without code. Start a new workflow with KLK8.
Results found
Linked to
| Label | Description |
|
|---|---|---|---|
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS | |||
A Gene Set from LINCS |
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
A Gene Set from LINCS
